★ Mounjaro demonstrated superior mean HbA1c Reductions with with every dose, outperforming comparators in all phase 3 studies
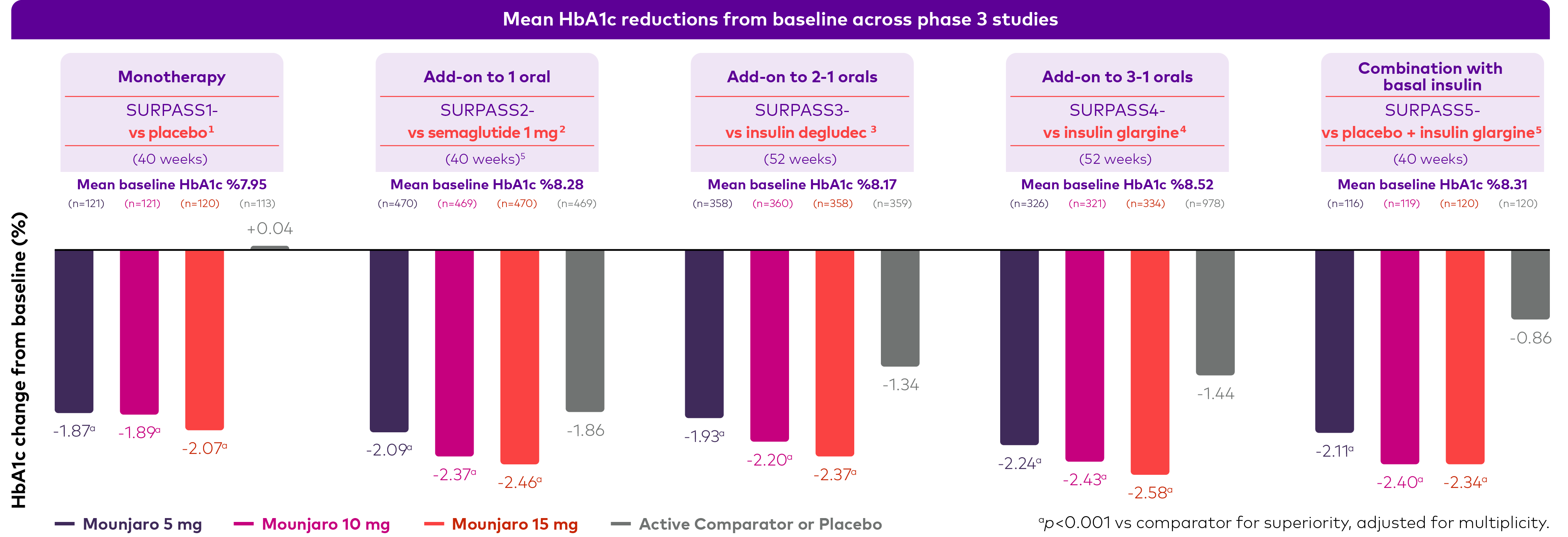
1- Rosenstock J, et.al, Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.9.
2- Frías JP, Davies MJ, Rosenstock J, et al. SURPASS-2 Investigators. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.10.
3- Ludvik B, et.al, Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet. 2021;398(10300):583-598.11.
4- Del Prato S, et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. The Lancet. 2021 Nov 13;398(10313):1811-24.12.
5- Dahl D, et.al, Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA. 2022;327(6):534-545.
Mounjaro vs. Placebo
(T2D patients who had inadequate glycemic control with diet and exercise)
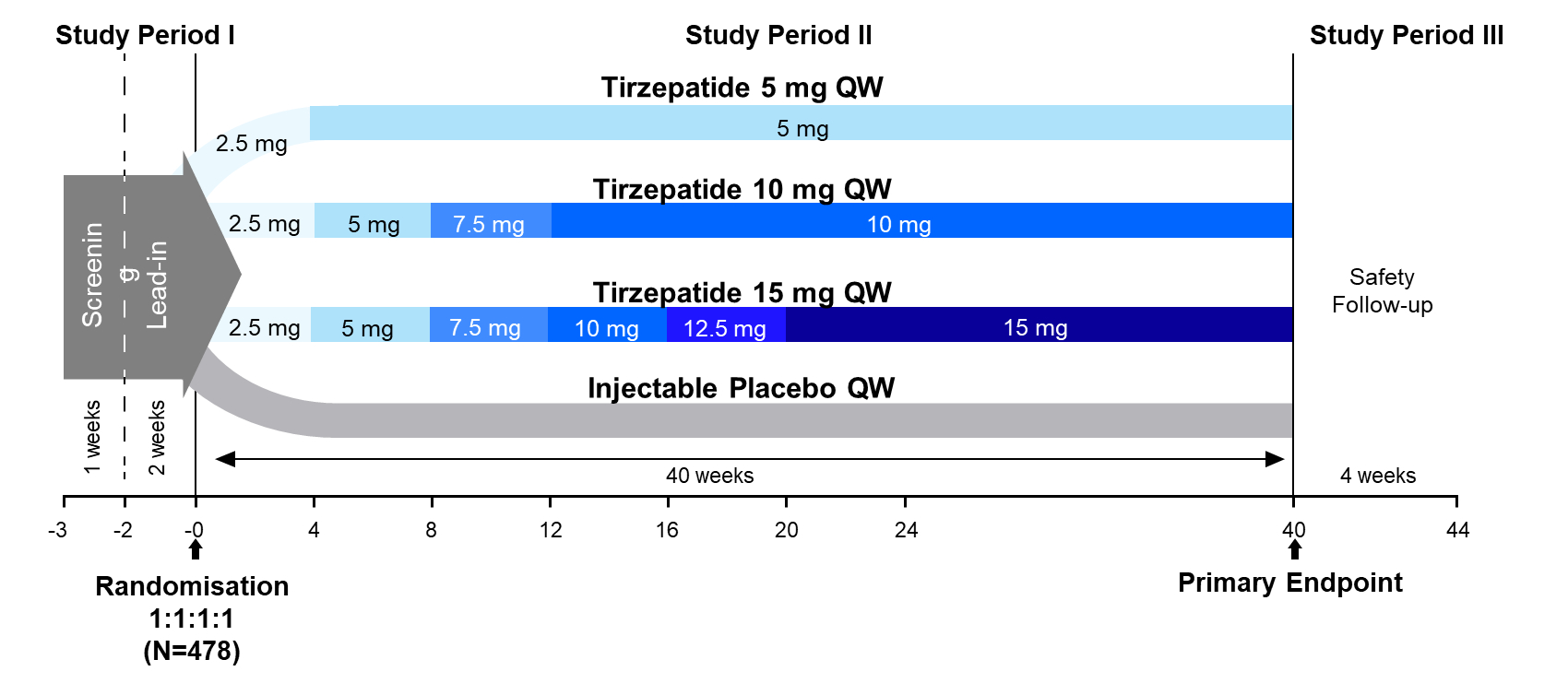
SURPASS-1 was a 40-week, double-blind, placebo-controlled, phase 3 trial that randomized 478 adult patients with T2D who had inadequate glycemic control with diet and exercise to receive once-weekly SC Mounjaro 5 mg, 10 mg, or 15mg, or placebo (1:1:1:1 ratio).
Study participants had a mean baseline A1C of 7.9% and mean T2D duration of 4.7 years.
PRIMARY ENDPOINTS
Superiority of Mounjaro 5 mg, 10 mg, and/or 15mg to placebo in mean change from baseline in A1C at 40 weeks.
KEY SECONDARY ENDPOINTS
Mean change from baseline in fasting serum glucose to week 40.
The proportion of participants with HbA1c target values of less than 7∙0% (<53 mmol/mol) and less than 5∙7% (<3 mmol/mol)
Mean change from baseline in bodyweight.
★ Mounjaro demonstrated SUPERIORITY in HbA1c reduction vs. placebo
PRIMARY ENDPOINTS
Mean Change in HbA1c from baseline to week 40
Mean Change in HbA1c from baseline to week 40
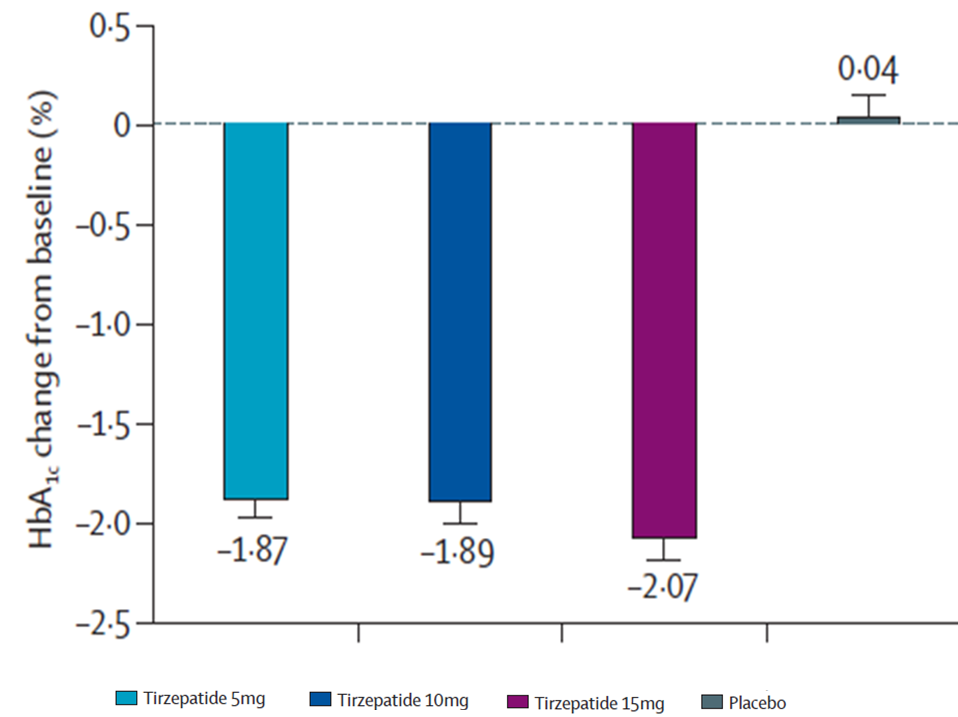
Significant Reductions
in HbA1c with Mounjaro 15mg at 40 weeks
★ Mounjaro demonstrated SUPERIORITY in HbA1c reduction vs. placebo
PRIMARY ENDPOINTS
- Mean Change in HbA1c from baseline to week 40
Mean Change in HbA1c from baseline to week 40
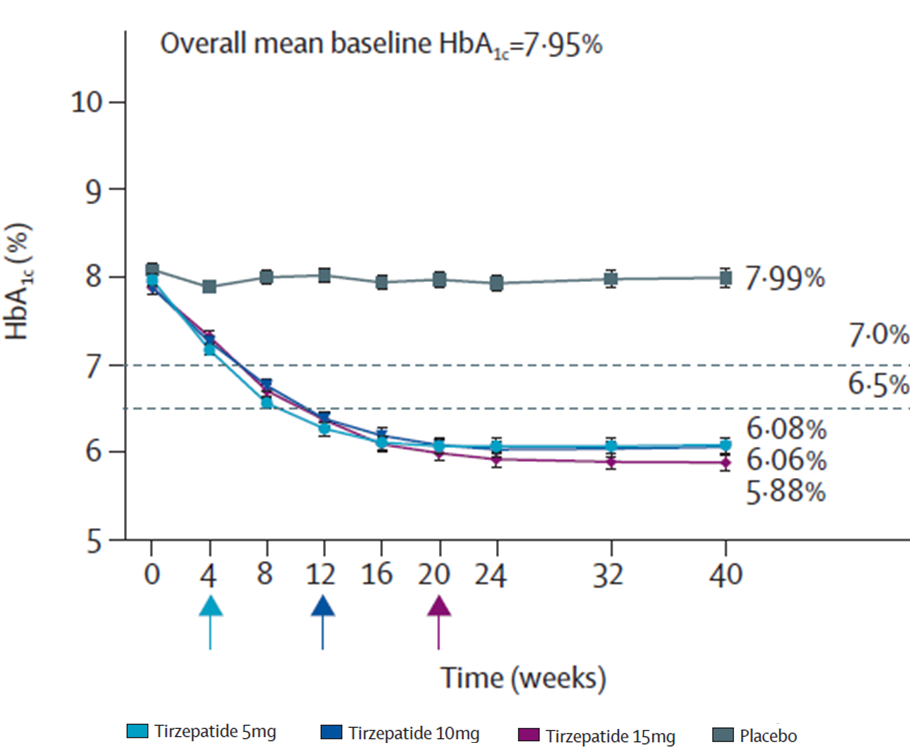
in HbA1c with Mounjaro at 40 weeks
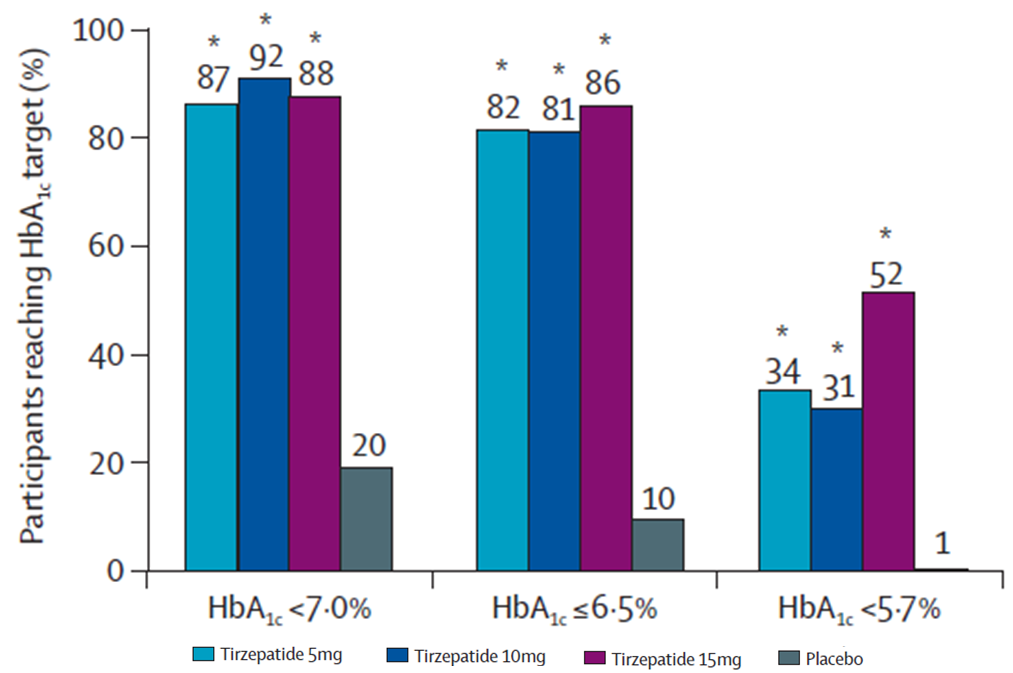
★ A significantly higher proportion of participants given Mounjaro reached HbA1c targets (<7·0%, ≤6·5%, and <5·7%)
Percentage of participants reaching HbA1c targets (<7·0%, ≤6·5%, and <5·7%)
★ Significant Weight Reductions with Mounjaro vs. Placebo
Change from baseline in Bodyweight at 40 weeks
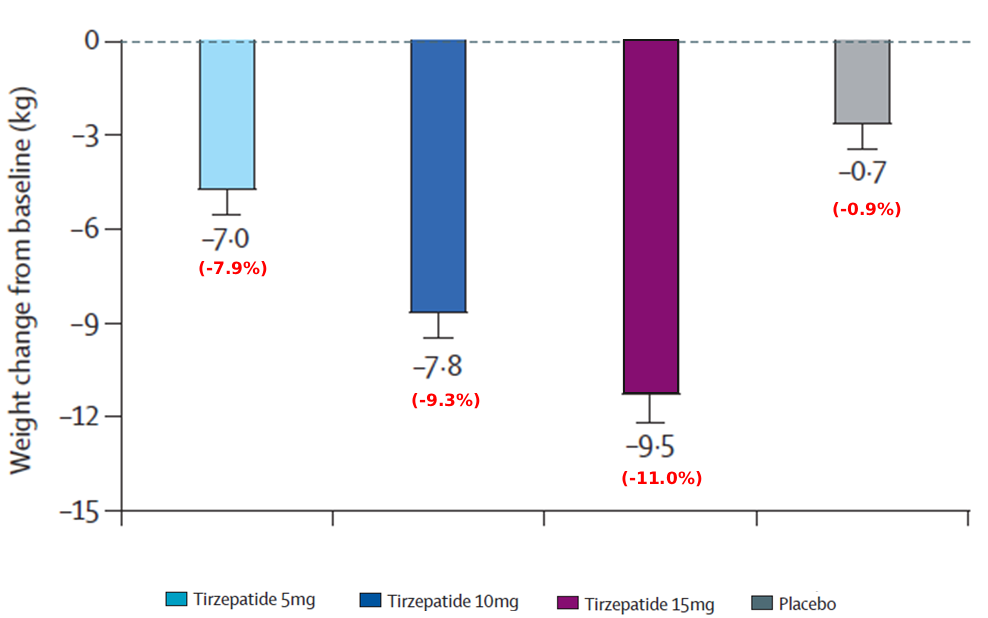
in bodyweight with Mounjaro 15mg at 40 weeks
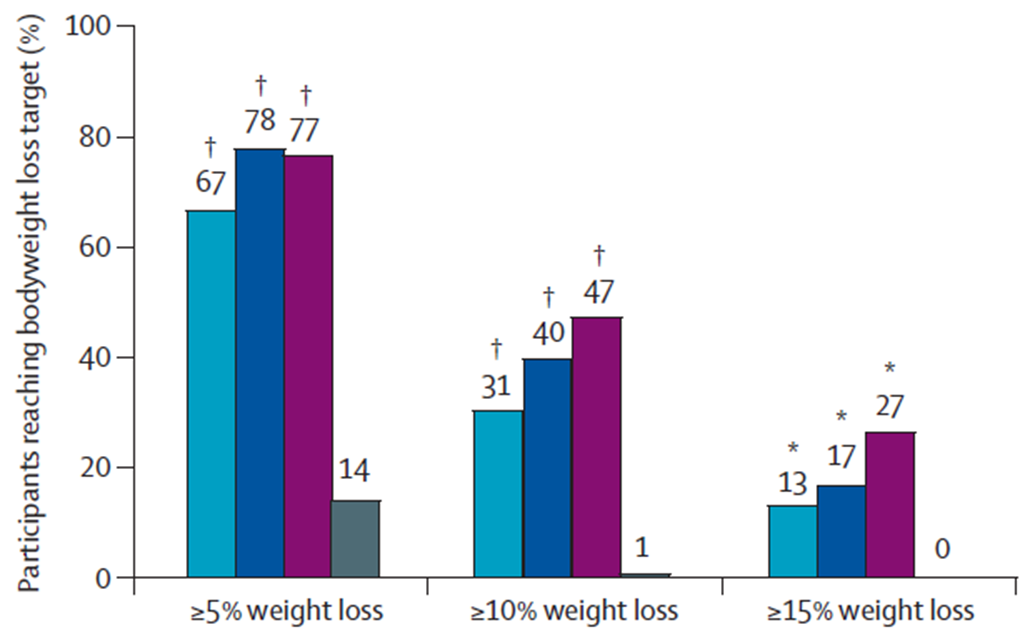
★ A significantly higher proportion of participants given Mounjaro reached weight loss aims (≤5%, ≤10%, and ≤15%)
Percentage of people who achieved ≥5%, ≥10% and ≥15% Weight Reductions at 40 weeks
Rosenstock J, et.al, Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.